Chapter 12: Type II Hypersensitivity Reaction and ADCC (Cytotoxic reaction)
Type II HYPERSENSITIVITY REACTION, ADCC (CYTOTOXIC REACTION)
Definition
In type II, the target is fixed in tissue or on the cell surface. This is mediated by IgG or IgM binding to the specific cell or tissue. The damage will be limited to the tissue or the cells where this reaction will take place.
Antigen: It is present or is a part of the cell membrane. It may be:
- Exogenous Ag: Microbes, parasites, drugs.
- Intrinsic Ag: Autoimmune diseases, and these are self-Ag.
Antibody: This is mainly due to IgG and occasionally IgM. Rarely IgA can give this reaction.
The effector cells are macrophages, neutrophils, eosinophils, and NK (natural killer) cells.
Mechanism
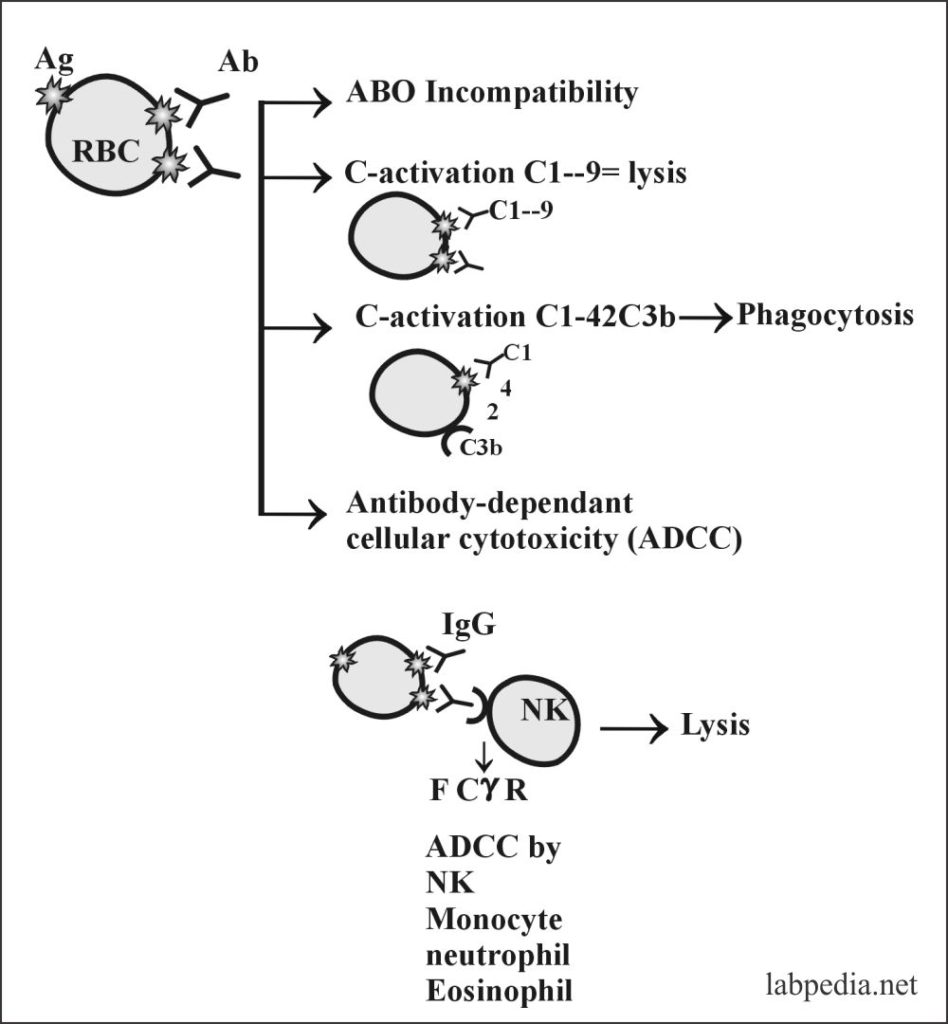
- Complement activation:
- The antibody attaches to Antigen on the surface of cells and activates the complement system, which leads to lysis.
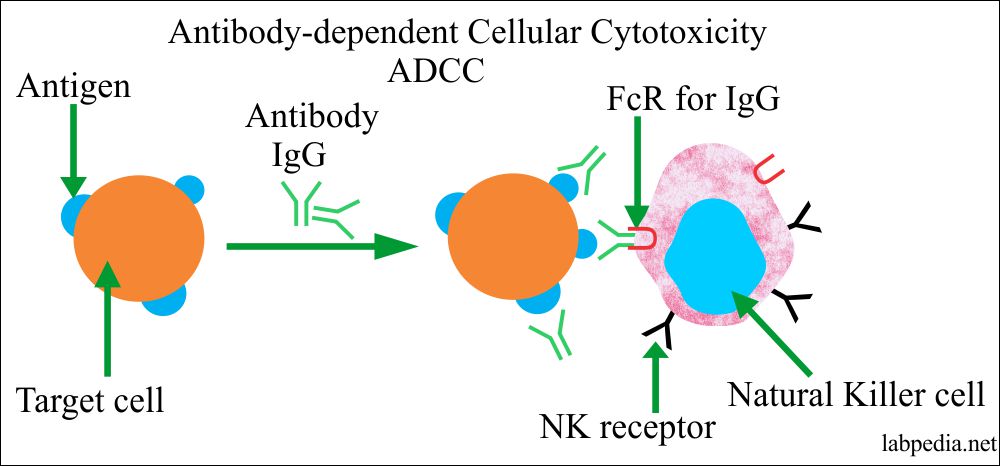
- Antibody-dependent cellular toxicity (ADCC):
- Sometimes Antibody is attached to Antigen on the cell surface will bring this complex near to NK cells or other phagocytic cells possessing the Fc-Receptor and leads to antibody dependant cellular cytotoxicity (ADCC).
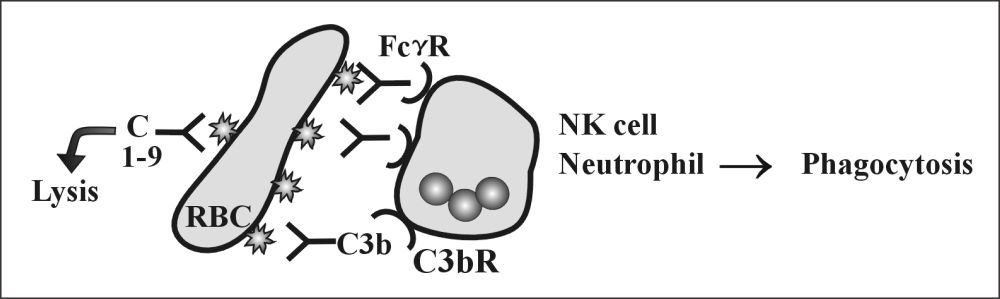
- Opsonization and phagocytosis:
- The antibody binds to an antigen and makes it a target for phagocytosis, and this process is called opsonization.
There are the following examples of Type II Reactions in our human body, leading to various diseases:
- RBC-Hemolysis
- Blood transfusion reactions: This may take place because of mismatched blood transfusion.
- Hemolytic diseases of the newborn (HDN), e.g., Rh-incompatibility and ABO-Incompatibility.
- Autoimmune hemolytic anemia (AHA). This takes place due to the loss of immunologic tolerance.
- It can be divided into three main types:
- Warm-reactive autoantibody, these autoantibodies react with antigen at 37 °C.
- Cold reactive autoantibodies react with antigen below 37 °C and mostly at 4 °C.
- These antibodies are provoked by the drugs causing an allergic reaction.
- It can be divided into three main types:
The possibilities of RBC Ag and Ab reactions are:
-
- Agglutination.
- Complement dependant lysis.
- Phagocytosis.
Major blood groups involved in transfusion reactions are:
Blood group type Chromosomes location Antigen on RBC Phenotypes of blood groups ABO 9q34.2 - Antigen A
- Antigen B
- O has no antigen
- A = 42%
- B = 8%
- AB = 3%
- O = 47%
Rhesus 2 closely linked loci = Major antigen is RhD
1p36.11
- C or c
- D or d
- E or e
- RhD+ = 85%
- RhD– = 15%
Duffy 1q23.2 - Fya
- Fyb
- Fy
- Fya+b+ =46%
- Fya+ b- = 20%
- Fyb+ a- = 34%
- Fya-b- = 0.1%
Kell 7q34 K or k - Kk = 8.8%
- kk = 91%
- KK = 0.2
MN Located on chromosomes 4
(46 antigens)
4q31.22
- M
- N
- S and s
- MM = 28%
- MN = 50%
NN = 27%
- Neutrophils: – There may be the destruction of neutrophils in SLE or due to drugs.
- Autoantibodies to the neutrophilic cytoplasmic antigen (ANCAs) are associated with several diseases.
- Antibody to proteinase-3, a cytoplasmic antigen (C-ANCA), is seen in Wegener’s granulomatosis.
- Antibodies to myeloperoxidase are seen more commonly in SLE and are located in the perinuclear granules (P-ANCA).
- Antibodies to P-ANCAs are seen in glomerulonephritis and vasculitis.
- These autoantibodies are generally neutrophil-specific, and these antibodies can be detected by immunofluorescence stain.
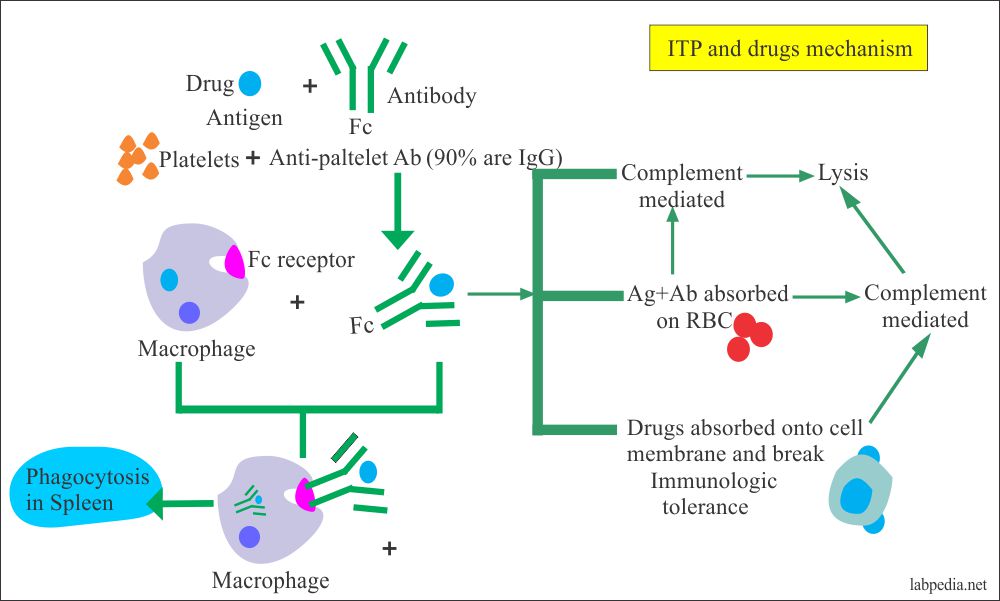
- Platelets: – The platelets are destroyed in ITP(idiopathic thrombocytopenic purpura) and drugs like sedormid and quinidine.
- Autoantibodies to platelets are seen in 70% of the cases of ITP.
- It is increased removal of platelets from the circulation; this is mediated primarily by the splenic macrophages.
- Mechanism:
- The removal is by the immune adherence receptors on these cells.
- This condition mostly develops after bacterial or viral infection.
- This may be associated with autoimmune diseases like SLE.
- Lymphocytes: – Lymphopenia may be seen in SLE.
- Kidney: – Type II reaction gives anti-glomerular-basement membrane nephritis (anti-GBM nephritis) and Good Pasteur’s syndrome.
- There are antibodies against the collagen type IV, which is the major component of the glomeruli’s basement membrane.
- In 50% of the cases, it binds the complement and is usually IgG.
- Microscopically there is severe necrosis of the glomeruli, with fibrin deposition.
- The Goodpasteur first noticed this along with lung hemorrhage, so this was given his name Goodpasture’s syndrome.
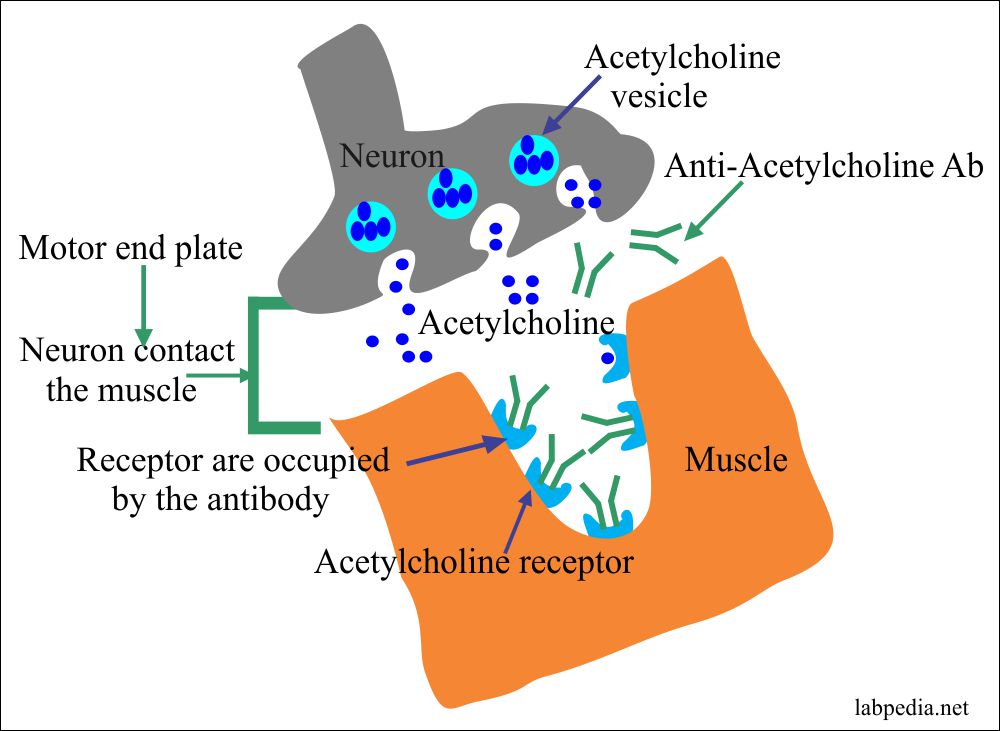
- Myasthenia Gravis:
- There is an extreme weakness in the muscles.
- There are autoantibodies against the acetylcholine receptor present on the surface of the muscle membrane.
- The acetylcholine receptors are present at the motor endplate, where the neuron contacts the muscles.
- Where Ab reacts with acetylcholine receptors.
- There is the transient weakness of the newborn babies because of the mother’s passive transfer (myasthenia gravis patient) IgG antibodies.
- Skin diseases:
- In pemphigus, there is a severe blister of the skin. There may be involvement of the mucous membrane.
- Pemphigus is strongly associated with a rare haplotype of HLA-DR4.
- Pemphigus Vulgaris, in this case, Antibodies are against the dermo-epidermal junction.
- Autoantibodies are against desmoglein-1 and desmoglein-3; these are components of the desmosomes.
- anti-desmoglein-3 tend to show mucosal disease, while with anti-desmoglein-1 and 3 will affect the skin and mucosa.
- The antibodies disrupt the cellular adhesion, leading to the breakdown of the epidermis.
- Drugs:- Alpha methyldopa (antihypertensive drug) gives hemolytic anemia. This drug acts as a hapten. Other drugs are phenacetin, amidopyrine, penicillin, and chlorpromazine.
- The mechanism may be:
- The drugs or their metabolites provoke a hypersensitivity reaction.
- The drugs may bind to the cell membrane as Alpha-methyl dopa attaches to the RBC membrane.
- The drugs-antibody complex is adsorbed on to the RBC membrane.
- Drugs may produce autoantibodies directed against the RBC antigens on themselves.
- The mechanism may be:
- Infectious agents like salmonella infection and tuberculosis.
Hemolytic Diseases of Newborn
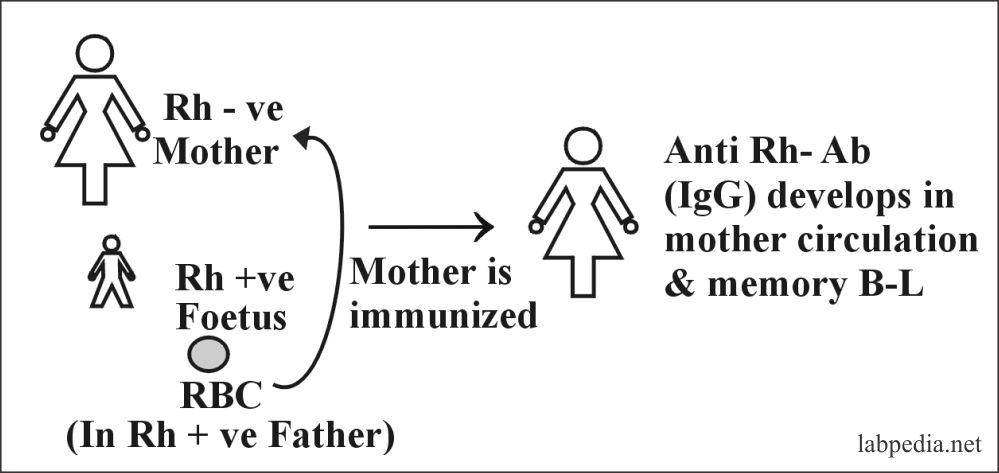
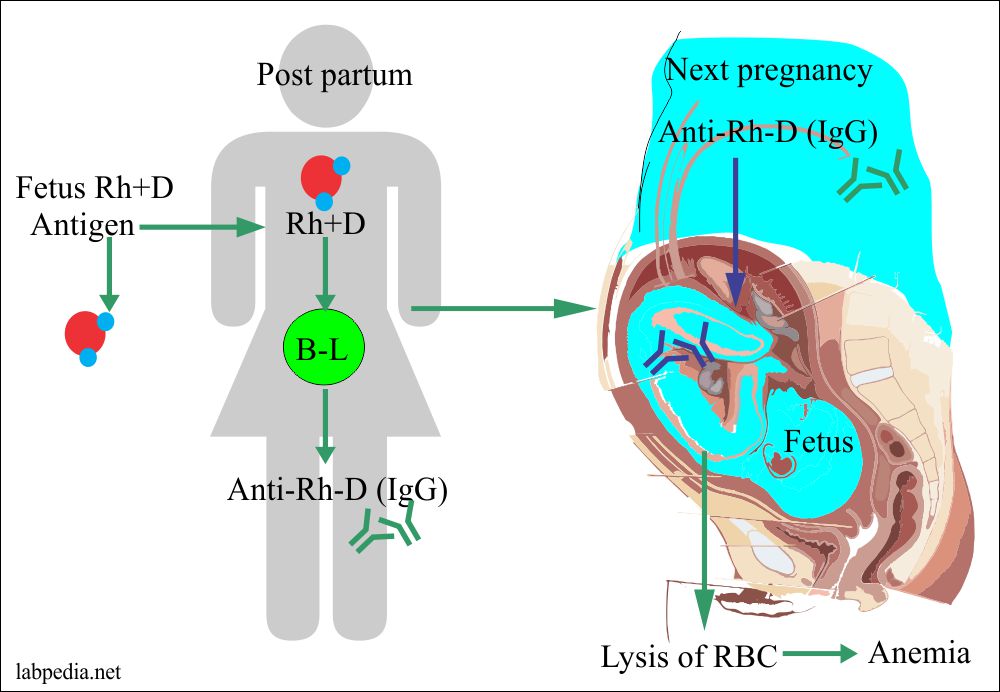
Rh-incompatibility
This is also called erythroblastosis fetalis or Hydrops foetalis. It is seen in 15% whites and 7% blacks.
This is because of the sensitization of Rh-negative mother by Rh +ve fetus, where the father is Rh +ve. Sensitivity is particularly to D-antigen (90%).
D-Ag is more potent; first, there is IgM anti-D formation followed by IgG anti-D. Anti-Rh Ab in mother appears after 6 weeks. This Ab leads to phagocytosis and hemolysis of fetal RBC due to the activation of the complement system because this IgG type Ab can cross the placental barrier.
Presensitization of the mother may occur due to the previous history of abortion or stillbirth.
Other blood groups leading to hemolytic disease of newborn are:
- Kell system due to K antigen.
- Reaction due to anti-K is much less common than the reaction to the Rh-D antigen because of the K-antigen’s low antigenicity.
- ABO blood group.
Signs and Symptoms of Rh-IgG antibody:
These are due to hemolytic anemia, which are:
- Anemia causes low oxygen-carrying capacity and leads to heart failure.
- An increase in bilirubin formation due to hemolysis causes jaundice and may lead to kernicterus.
- There is a hypoxic injury to the liver, heart, and brain.
- There is a low protein (only 2.0 G/dl), leading to generalized edema (Anasarca) or Hydrops foetalis.
- Jaundice appears within 24 hours in the newborn.
Low weight babies and premature newborns are at greater risk as these babies conjugating, and excretory systems of the liver are not fully developed.
2% of the fetus may sensitize the mother within 28-34 weeks of gestation, and these mothers are at more risk to develop Ab so needs anti-IgG (RhoGAM) in the last trimester.
Pathogenesis
- The First Rh+ve baby of Rh+ve father sensitizes the mother when his/her blood goes into Rh-negative Mother during the last trimester of the pregnancy when the cytotrophoblast is no longer present a barrier or during childbirth itself.
- The amount of blood may be more than 1.0 ml.
- If the mother and fetus’s blood groups are different, then there are chances that the mother may not be sensitized.
- In the case of compatible blood of mother and fetus, fetal RBC Rh +ve will survive and sensitize the mother. This first baby will escape hemolytic anemia.
- But the second baby will be the victim of anti-Rh Ab present in the mother circulation.
- First, IgM Ab forms and then IgG.
- These IgG type Ab can cross the placental barrier and goes into the fetal circulation, and causes hemolytic anemia.
- IF the father genotype is Rh+ve /Rh+ve, = all children are Rh+ve
- If father genotype is Rh+ve /Rh-ve = then 50% are Rh+ve.
Treatment
- Rh-positive baby mother should be given RhoGAM (anti-IgG) within 24-72 hours after the baby’s birth if delayed then there is no value or use.
- By giving the anti-IgG (Rhogam) decreased the number of HDN due to Rh-antibodies.
- Case due to other blood groups like Kell and ABO are increasing.
Diagnosis
The diagnosis of Ab in the mother can be detected by the indirect Combs test. For to find Rh antibodies in the baby, a direct Coombs test may be done.
ABO Incompatibility
Incidence
It is quite common and is seen in 20-25% of pregnant women. Only 10% of newborn develops hemolytic anemia and only 1/200 needs treatments.
Pathogenesis
Most Abs is IgM against Ag-A and Ag-B, and these can not cross the placental barrier. Due to unknown reasons, only O mother develops IgG type Ab against A and B Ag, without previous presentation. So may see reaction even in the first baby.
Fortunately, lysis of fetal RBC is less severe due to poor expression of Ag A and B on RBC and also due to the presence of Ag-A and B on other cells than only RBC, e.g. epithelial cells.
The only fetus with strong Ag-A and B shows evidence of hemolytic anemia.
Treatment
There is no method of protection.
The evaluation of the hemolytic anemia of newborn (HDN):
The following tests are advised prenatal or postnatal for the diagnosis of HDN.
- Blood grouping for ABO typing.
- Rh blood group typing.
- Screening of the alloantibodies (these antibodies target non-self RBC antigen).
- Check prenatal mother by amniocentesis.
- Check serum bilirubin of cord or newborn baby.
- Advise Coombs’ direct test on cord blood or newborn baby blood.
- Advise peripheral blood smear.
- Check for the Du in the blood.
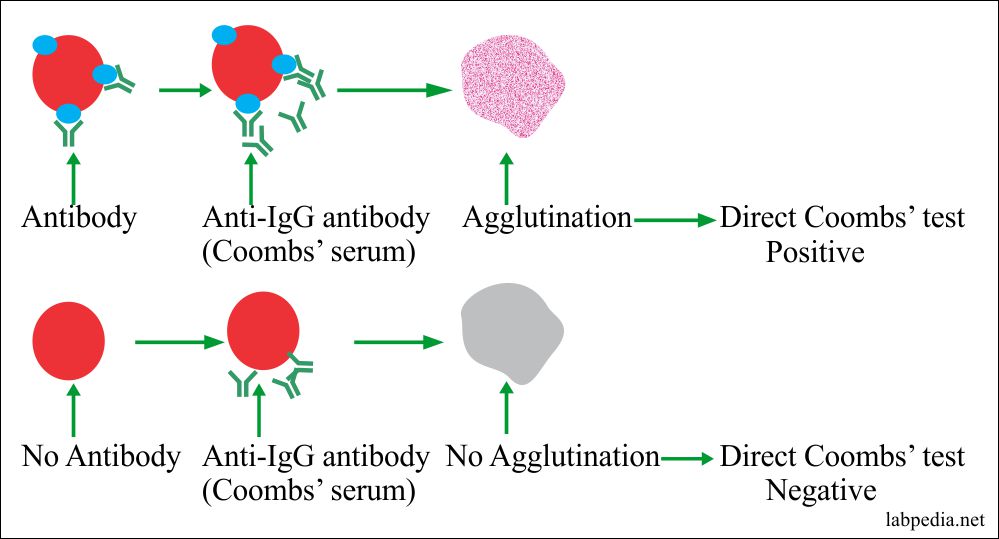
Coombs Test (Anti-Human Globulin Test)
This was first described by Morchu in 1908 and later on rediscovered in 1945 by Coomb, Morchu, and Race.
Purpose
- Evaluation of hemolytic anemia of the newborn.
- Red cell typing in blood banks.
- Diagnosis of autoimmune hemolytic anemia.
- Drugs induced hemolytic anemia.
This test may be done as:
Direct:- When Abs are present on cells or Ab-coated RBC, e.g. baby blood.
Indirect:- When Abs are free in the serum, e.g. in mother serum.
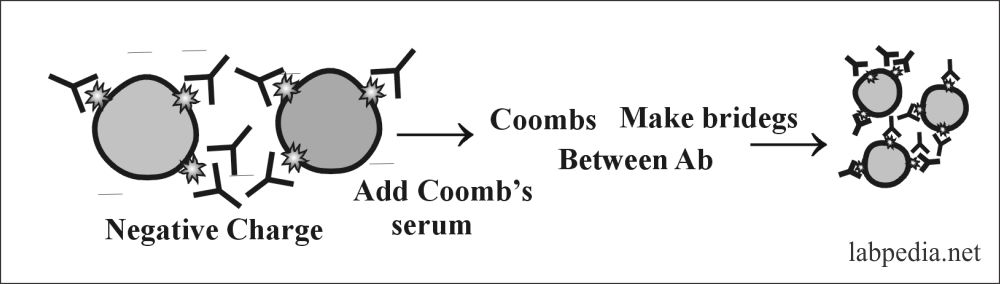
Principle
The RBCs have a negative charge, so IgG-Ab can not show agglutination. To overcome this negative charge and to see agglutination following means can be used:
- Addition of albumin(albumin phase).
- Addition of combs serum(Coombs phase).
- Addition of enzymes (enzyme phase).
- Coombs serum prepared in rabbit by injecting IgG and will get anti-IgG or Ab against Ab ( anti-anti-Ab).
Albumin will neutralize the negative charges and show agglutination. Coomb’s serum anti-IgG Ab form bridging between two Abs and show clumping.
Hypersensitivity reactions and immunologic mechanism of tissue destruction:
| Hypersensitivity reaction | Type of reaction | Time of development | Type of Antibody | Type of cells involved | Role of complement | Example of the disease |
| Type 1 | IgE dependent reaction | Immediate | IgE (by activation of B-lymphocytes) | Mast cells | No |
|
| Type II |
|
Immediate | IgG and IgM (by activation of B-lymphocytes) | Macrophagic cells | Frequently involved |
|
| Type III | Immune-complex mediated reaction | Immediate | IgG and IgM (by activation of B-lymphocytes) | Neutrophils | Yes |
|
| Type IV | Cell-mediated reaction | Delayed | None (by activation of the T-lymphocytes) |
|
None |
|


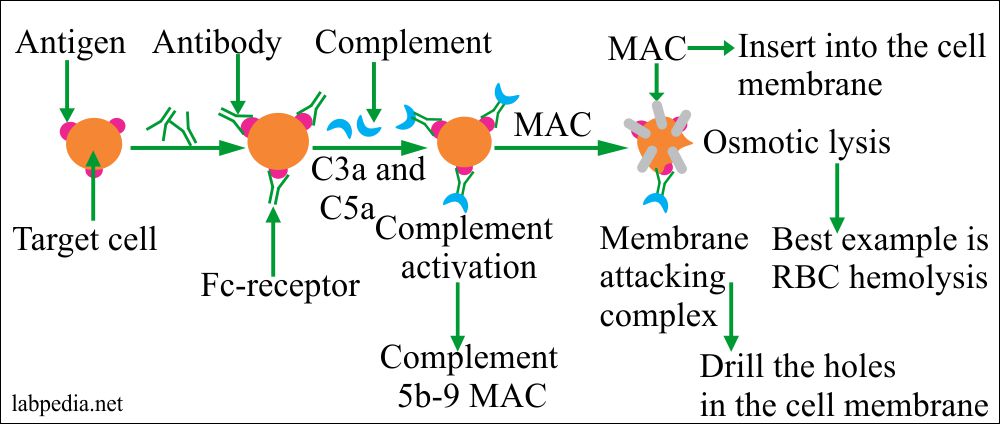
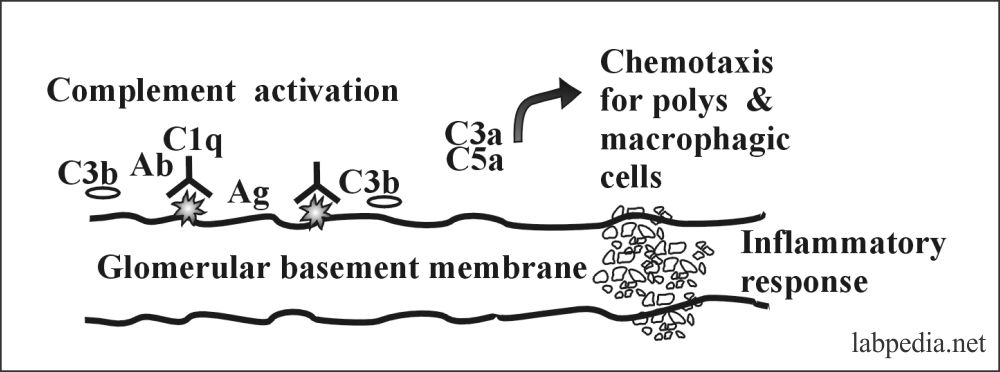

role of complement system in typeII hypersensitivity
amin
It is explained in the chapter. Mainly Complement will lead to lysis.