Ketoacidosis diagnosis, Diabetic Ketoacidosis
Ketoacidosis diagnosis
What sample is needed for the diagnosis of Ketoacidosis?
- Blood is needed to prepare the serum.
- A urine sample may also be used.
What are the indications for Ketoacidosis?
- Ketone bodies are advised in patients with diabetes mellitus.
- The advice in case of uncontrolled diabetes mellitus.
- Ketoacidosis is associated with alcoholism, starvation, and a high-protein diet.
How will you define Ketoacidosis?
- There is inhibition of glycolysis in ketoacidosis while glycogenolysis and gluconeogenesis are stimulated.
- There is hyperglycemia, deranged acid balance, and electrolyte imbalance.
- It is acute life-threatening metabolic acidosis due to uncontrolled diabetes mellitus ( usually type 1 and infrequently type 2).
- This condition occurs when decompensation by other diseases, insulin deficiency, and excess hormone-producing glucose.
What is another definition of Ketoacidosis?
- A lake of insulin leads to the metabolism of protein and fat to provide energy. Normally, energy is supplied by carbohydrates.
- Ketone bodies and other metabolic acids accumulate in the blood.
- Accumulation of ketone bodies (acetone, acetoacetate, and β-hydroxybutyrate) is the principal feature of diabetic ketoacidosis.
- There is hyperglycemia. Ketones and glucose appear in the urine.
- There is hyperosmolarity.
- Low pH.
- Patients have lethargy and may go into a coma.
How will you discuss the Pathophysiology of Ketoacidosis?
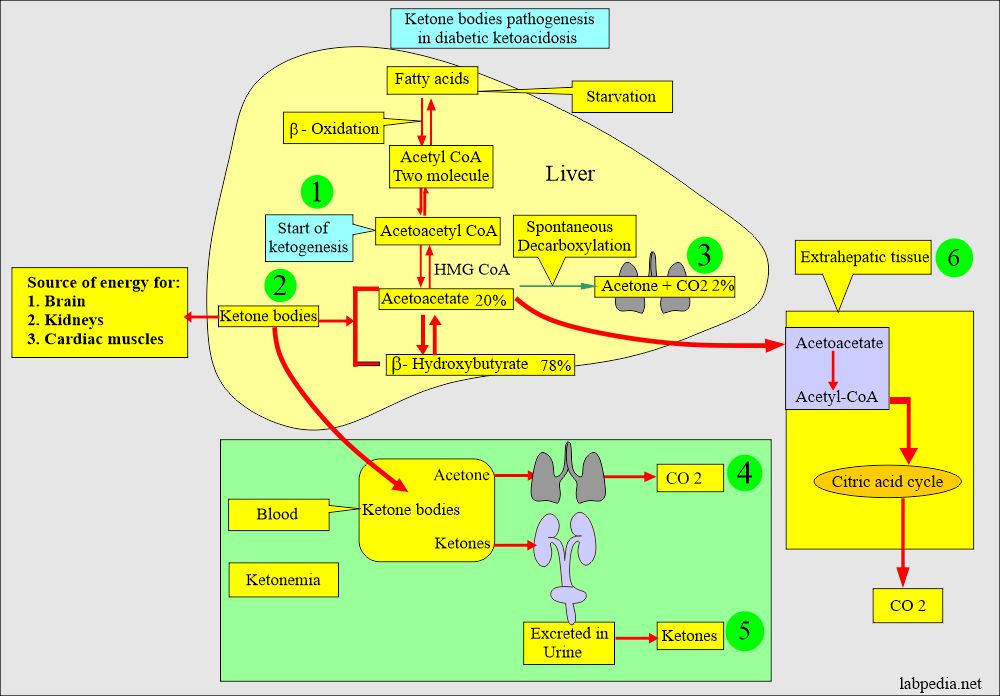
- Acetyl coenzyme A (acetyl CoA) from the TCA (Tricarboxylic acid cycle) is at the junction of glucose, protein, and fat metabolism.
- Free fatty acids enter the TCA cycle at Acetyl CoA. It changes to acetoacetyl CoA.
- It then forms HMG-CoA (3-hydroxy-3-methylglutaryl CoA).
- This HMG-CoA can be metabolized to cholesterol or converted to acetoacetate.
- Acetoacetate can change into:
- Spontaneous decarboxylation to acetone in the lungs.
- Or Enzymatic reduction to β-hydroxybutyrate.
- Acetoacetate and β-hydroxybutyrate are called keto acids or ketone bodies.
- Ket acids are the energy source for the brain, kidneys, and cardiac muscles.
- Kidneys excrete an excess of Acetoacetate and β-hydroxybutyrate with a loss of Na+ and K+.
- Loss of Na+ and K+ from kidneys leads to retention of the H+ ions.
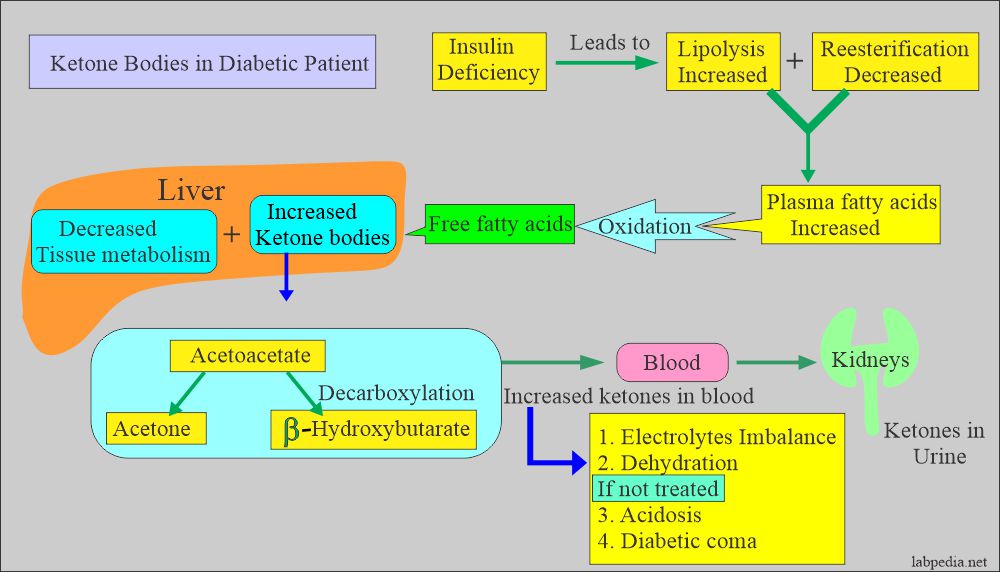
- In diabetes mellitus, low insulin levels mobilize fatty acids from triglycerides.
- Fatty acid degradation increases as it becomes the cell’s major energy source.
- Increased fatty acid metabolism gives rise to increased molecules of acetyl CoA.
- This excess Acetyl CoA enters the TCA cycle to produce energy.
- Still, some of it forms ketone bodies and ketosis (keto acids).
- Increased production of keto acids utilizes HCO3– and lowers the blood pH (acidosis).
- This mechanism occurs in starvation, except there is hypoglycemia instead of hyperglycemia.
- The liver takes up long-chain fatty acids.
- In the liver, they are reesterified to triglycerides and stored.
- Or these are converted to very low-density lipoprotein and return to the blood.
- Infants have earlier ketonuria than adults after decreased food intake.
What are the findings in uncontrolled diabetes mellitus?
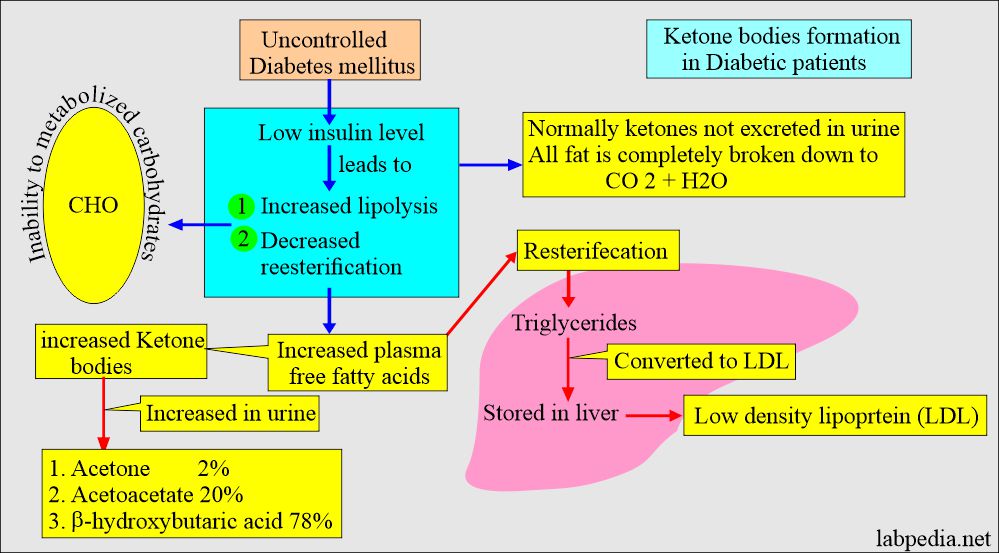
- Low insulin level leads to:
- Increased lipolysis.
- Decreased reesterification.
- There are increased plasma-free fatty acids.
- Insulin and glucagon ratio changes.
- This ratio is changed and enhances fatty acid oxidation.
- It leads to increased ketone body formation in the liver and decreased peripheral tissue metabolism, which leads to blood accumulation.
- There is an accumulation of acetoacetate ketone bodies in the blood.
What are the signs and symptoms of diabetic ketoacidosis?
There may be :
- A cough.
- Fever.
- Chills.
- Chest pain.
- Arthralgia.
- Dyspnoea.
- Tachycardia.
- Hypotension.
- Hypothermia.
What are the causes of Ketone Bodies Formation?
- Starvation.
- Frequent vomiting.
- Diabetes mellitus.
- Glycogen storage disease (von Gierke disease).
- During pregnancy.
How will you diagnose diabetic ketoacidosis?
- The glucose level may range from 300 to 500 mg/dL.
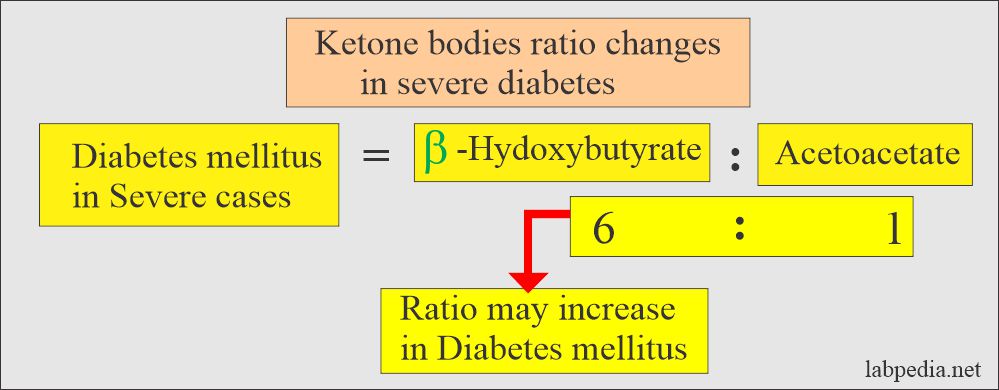
- Ketone bodies are beta-hydroxybutyric acid, acetoacetic acid, and acetone.
- These are produced during gluconeogenesis due to increased fatty acid oxidation to acetyl CoA.
- An excessive concentration of ketone bodies in the blood is called ketonemia.
- When there is increased excretion in the urine, it is called ketonuria.
- Electrolytes show :
- Decreased Sodium.
- Increased Potassium.
- The anion gap is increased.
- Total CO2 decreased.
- Blood gases show:
- pH decreased.
- Metabolic acidosis.
- Bicarbonates are decreased.
What are the differentiating points of Ketoacidosis (Diabetic ketosis)?
| Test | Lab findings/Clinical presentation |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
How will you detect ketone bodies in the urine (Ketonuria)?
Discuss sodium nitroprusside reaction?
- This reaction gives lavender color.
- This is very sensitive, ten times more for acetoacetate than with acetone. It detects acetoacetate and acetone.
- It does not react with β-hydroxybutyrate.
How will the Sodium nitroprusside react with ketone bodies?
- Take 0.5 to 1.0 grams (pea-size) of well-mixed nitroprusside reagent in a test tube or on the tile.
- Add a drop of a fresh urine sample to the reagent (Nitroprusside).
- After one minute, check the color.
- Results reported as:
- No color change = Ketones negative.
- Slight purple color = Ketone positive (+).
- Moderate purple color = Ketone positive (++).
- Dark purple color = Ketone positive (+++).
Questions and answers:
Question 1: How will you diagnose ketone bodies?
Question 2: What will be the pH level in diabetic ketoacidosis?